September 3, 2024
The Ultimate Guide To Peptides In Muscle Building: Advantages, Types, And Threats
Anabolic Steroids And Other Appearance And Performance Enhancing Medicines Apeds National Institute On Drug Abuse Nida

A Mechanism Of Action Of Peptides In Muscle Development And Advancement
Due to the fact that suckling differs significantly from adult feeding actions, a number of previous studies have used designs of adult-like independent intake to study the ontogeny of food consumption controls in pups (30-- 33). These researches have actually shown that, before P6, food consumption is mainly prevented by gastric fill (34, 35), and, by P9, independent consumption can be inhibited by nutritive signals (1, 36). In contrast, nutritious signals do not appear to prevent suckling up until a minimum of P14 (35, 37). We, nonetheless, observed MTII-mediated restraint of milk consumption in suckling dogs at all ages researched, from P6 to P16. This restraint therefore does not appear to reflect the developing progression of repressive ingestive controls but rather most likely shows activation of central melanocortin receptors that are already present at birth. Importantly, these research studies demonstrate that, not only does MTII prevent solid food intake in adult rats, but it can hinder suckling-mediated milk intake as very early as P6, a time when food consumption is largely mediated by stomach fill (1 ).
What Concerning Afamelanotide?
The mean worth per region per pet was established and made use of for statistical analysis. It's known for promoting fat loss and muscle growth over a prolonged period. Unlike other peptides requiring constant application due to short half-lives, CJC-1295 is specifically advantageous for those seeking less regular application.
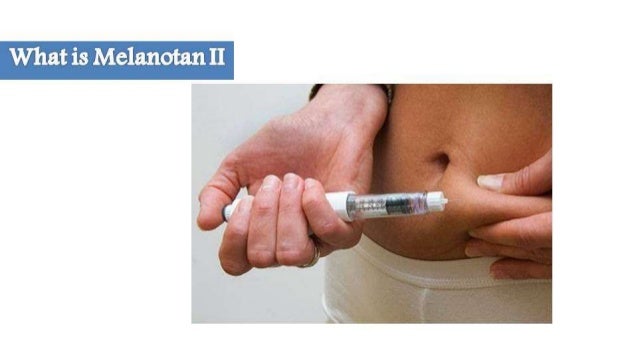
Melanotan Recommendations
Let your treatment group recognize you made use of tanning nasal spray so they can much better identify and treat any type of damaging reactions to melanotan. If you use a tanning nasal spray, anything more than moderate adverse effects warrants a visit to a medical care professional. In addition to the safety elements, the nasal sprays having melanotan II aren't going to give people the year-round tan they could anticipate.
- Afamelanotide has been revealed with Stage III researches to lower phototoxic reactions and recovery time in patients with erythropoietic protoporphyria.
- A similar tachyphylactic response to persistent MTII management has been observed in adult rats (21, 22) and might be attributable in part to lowered circulating leptin levels or other additional impacts of decreased power intake.
- It's renowned for promoting the pituitary gland to launch growth hormonal agent, which plays a critical role in muscle development and fat loss.
- We hypothesize that these short-term NPY populations drive food consumption before the facility of ARH feeding neurocircuitry and/or promotes the shift to independent ingestion.
Food and Medicine Administration-recommended restrictions for exposure to such alarmingly high radiation Anabolic steroids act at androgen receptors to influence cellular functioning and gene expression. Non-steroidal anabolics, include insulin, insulin-like growth hormonal agent (IGF), and human growth hormonal agent (HGH)-- compounds that are produced by the body and are recommended for genuine clinical usages however also sometimes misused for efficiency improvement. Though afamelanotide is similar to melanotan, the FDA has actually not accepted it for use as a self-tanner. The FDA approved this drug in 2019 as a therapy for adults with erythropoietic protoporphyria (EPP). Tanning hair salons advertise that getting a "controlled" tan in a tanning bed at the beginning of the summertime protects you by making it harder for you to shed when you go outside. The emergency and recommendation resources listed above are available to people situated in the United States and are not operated by the National Institute on Drug Abuse (NIDA). NIDA is a biomedical research study organization and does not supply personalized medical guidance, therapy, counseling, or legal consultation. Details offered by NIDA is not a replacement for professional https://seoneodev.blob.core.windows.net/pharma-warehousing/compounding-pharmacy/product/hustlers-make-use-of-barbie-trend-to-hock-illegal-nasal-spray.html treatment or legal consultation. Nutritional/dietary supplements are compounds bought legitimately from dietary shops or using the net that are frequently taken in combination with other APEDs. Philippe Wolgen, "Alpha-MSH derivatives for the treatment of photodermatoses" E.U.

Social Links